Which phase of matter is made up of particles that are packed closely together, with both a definite shape and a definite volume? When 13.6 grams of KCl are dissolved in enough water to create a 180-gram solution, what is the solution's concentration, expressed as a percent by mass? Organic compounds that contain only carbon and hydrogenĪccording to the collision theory and model created to explain the collision theory, what happens to a catalyst in a reaction? Which of the following correctly describes a hydrocarbon? Which of the following is the salt in the Brønsted-Lowry neutralization reaction? It increases the average kinetic energy and there are more collisions per minute. What term explains why some materials heat up more quickly than others?Īccording to the collision theory and model created to explain the collision theory, what is the best explanation for why a higher temperature makes a reaction go faster? 25 L of 12 M HCl solution to end up with 2.0 L of 1.5 M HClģ mol*1L/12M=.25L 1/4 of a liter of HCl is needed to go with 1.75L of water. How could you prepare the solution needed for the lab experiment? Show all the work used to find your answer.Īdd 1.75 L of H2O to. Heat from the Earth's core and pressure on the remains of dead plants and animals create which of the following resourcesĪ laboratory experiment requires 2.0 L of a 1.5 M solution of hydrochloric acid (HCl), but the only available HCl is a 12.0 M stock solution. How would a system in which both matter and energy are exchanged freely between the system and the surroundings be classified? On a potential energy diagram, what is true about activation energy?Īctivation energy is greater for endothermic reactions. What is the concentration of hydronium ions in a solution with a pH = 3.9? 
The chain reaction continues until all uranium-235 isotopes have split, or until the neutrons fail to collide with any more uranium-235 nuclei. Which of the following best describes a chain reaction in a nuclear fission reaction? The water and the metal's temperature will reach the same temperature. If you drop a 50 gram piece of metal that has a temperature of 110☌elsius into 1000 grams of water at 25☌elsius, what best describes what would occur? 
Plasma forms when a gas reaches extremely high temperatures, and electrons are released from the atoms. Which of the following best describes the formation of plasma? They move more slowly, resulting in the formation of a solid. 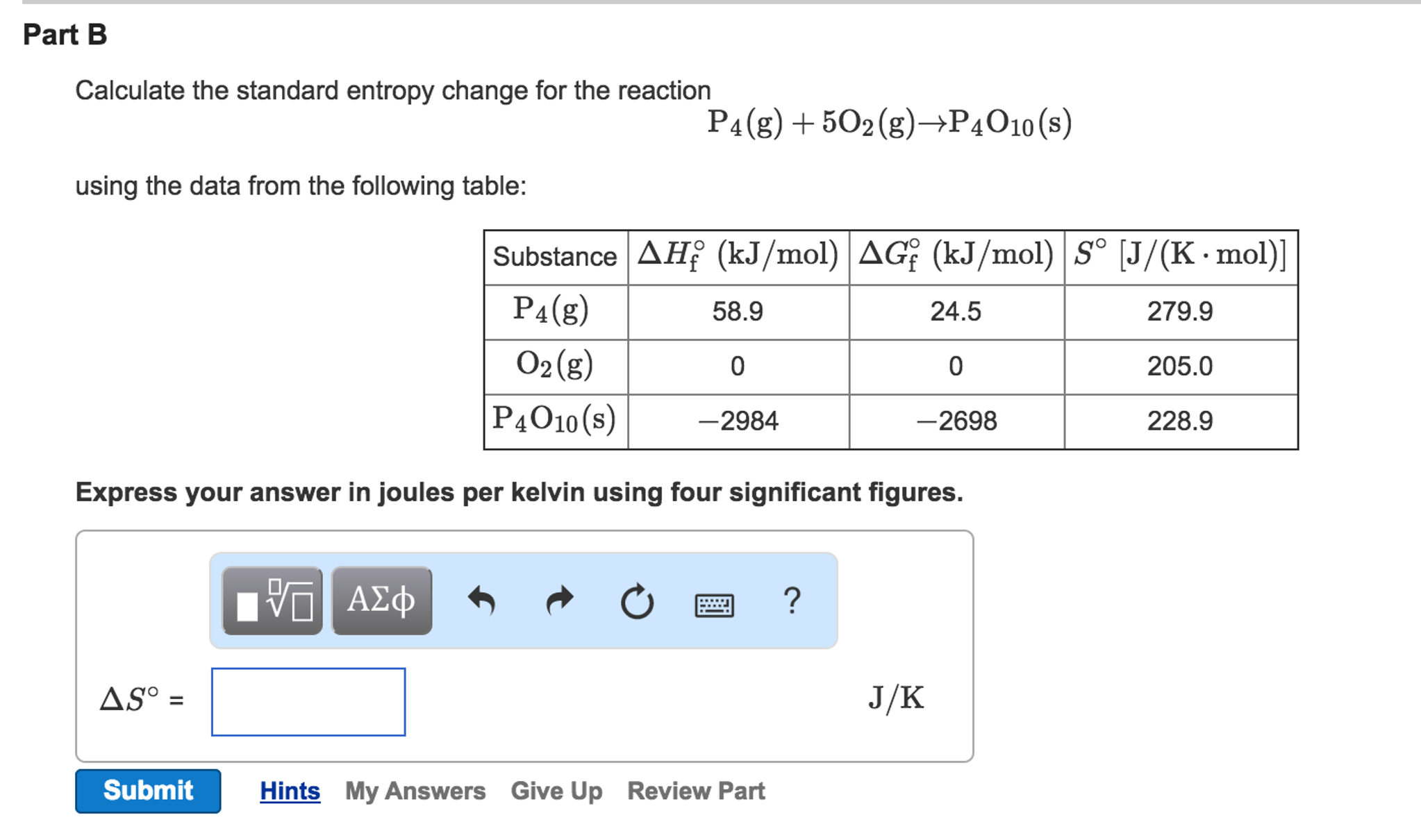
Which of the following best explains what happens when the kinetic energy of particles in a liquid state decreases? The particles have less kinetic energy in the liquid phase than they do in the gas phase. How does the kinetic energy of a substance's particles in the liquid phase compare to their kinetic energy in the gas phase? If 64 grams of O2 gas occupy a volume of 56 liters at a temperature or 25☌, what is the pressure of the gas, in atmospheres (atm)? How many liters of fluorine gas can react with 32.0 grams of sodium metal at standard temperature and pressure? Show all of the work used to find your answer.ģ2 g Na x (1 mol Na / 23.0 g Na) x (1 mol F2 / 2 mol Na) x (22.4 L / 1 mol F2) = 15.6 L of fluorine Which of the following describes what occurs as a gas becomes a liquid?Ī physical change occurs the particles of the gas vibrate more slowly and their kinetic energy decreases. Which of the following best describes this type of reaction? Hydrogen peroxide will decompose if the bottle is left open and the resulting release of energy will heat up the solution. Chemistry Final Final Chemistry Exam Question 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
